
The Late Whiplash Syndrome: A Psychophysical Study
Martina Moogc, John Quintnerb, Toby Halla and Max Zusmana
a School of Physiotherapy, Curtin University of Technology, Shenton Park WA 6008 (Australia), bWyllie Arthritis Centre, Shenton Park WA 6008 (Australia)
Corresponding Author: Martina Moog, University of Sydney, Pain Management and Research Centre, Royal North Shore Hospital, St. Leonards NSW 2065, Australia, Tel: +61-2-9926 7990, Facsimile: +61-2-9926 6548, Email: [email protected]
Category: Clinical article
Keywords: Whiplash injury, Allodynia, Secondary Hyperalgesia, Psychophysics, SCL-90-R
Abstract
Some patients who have sustained whiplash injuries present with chronic widespread pain and mechanical allodynia. This single blind, case control matched study of 43 chronic whiplash patients sought to examine psychophysical responses to non-noxious stimuli and their relationship to psychological profiles.
Symptom Check List-90-R, Neck Disability Index and Shortform McGill Questionnaire were completed prior to testing. Qualitative stimuli comprised light-touch, punctate pressure, moderate heat and cold. Additionally, sustained vibration was administered using a vibrameter which allowed ramping of either frequency or amplitude.
Twenty-eight patients reported vibration-induced pain. No control subject experienced pain in response to vibration. No significant differences in perception threshold to vibration were noted between patients and control group. Twenty-three patients and ten control subjects reported painful responses to cold. Eleven patients and nine control subjects experienced pain in response to moderate heat. Four patients rated punctate pressure and one patient rated light-touch as painful.
SCL-90-R profiles revealed an overall elevated level of distress in the whiplash group. No significant difference was found between patients with and without vibration-induced pain for any dimension of the SCL-90-R.
Pain in response to non-noxious stimulation over presumably healthy tissues suggests that central mechanisms are responsible for ongoing pain in at least some whiplash patients. The additional findings of pain on punctate pressure and hyperalgesic responses to heat and cold stimuli are consistent with enhanced central responsiveness to nociceptor input. These results have important therapeutic and prognostic implications.
Introduction
In Western societies motor vehicle insurance claims due to ‘whiplash-type’ injuries have been described as reaching ‘epidemic’ proportions (Spitzer et al., 1995; Ratcliff, 1997). Those who become chronically disabled impose a major social and financial burden upon society (Barnsley et al., 1994; Gargan et al., 1997, Ferrari and Russel, 1999a).
The pathogenesis of the so-called ‘late whiplash syndrome’ (Pearce, 1989) is poorly understood (Koelbaek Johansen et al., 1999). Some maintain that chronic pain and disability reflect the influence of such ‘environmental’ factors as management interventions by health care professionals and the medico-legal process (Schmand et al., 1998; Ferrari and Russell, 1999b; Obelieniene et al., 1999; Pearce 1999; Cassidy et al., 2000). Others argue that ongoing pain is likely to have a somatic basis and that any associated physical and psychological disability are secondary, in so far as they resolve coincidentally with pain relief (Radanov et al., 1995, Radanov et al. 1999; Wallis et al., 1998). Cervical zygapophyseal joints have been proposed as a possible anatomical source of chronic pain in at least 50% of these patients (Wallis et al., 1997).
Animal experimental studies have shown that tissue injury causing prolonged noxious input can produce functional changes within the central nervous system which are maintained long after nociceptive input has disappeared or has become minimal (Woolf and Doubell, 1994). This state, which is known as central sensitization, is manifested as an increased response or reduced threshold to afferent input, prolonged after-discharges to repeated stimulation, an expansion of the receptive fields of dorsal horn neurones and an increase in their spontaneous activity (Coderre et al., 1993; Devor, 1994; Koltzenburg et al., 1994).
The clinical correlates of central sensitization are likely to be hyperalgesia (an increased response to a noxious stimulus) (Merskey and Bogduk 1994), allodynia (pain in response to a non-noxious stimulus) (Merskey and Bogduk 1994), widespread pain, and stimulus-independent pain (Coderre et al., 1993). There is an emerging body of evidence derived from psychophysical testing which is consistent with an underlying state of hypersensitivity in the central nervous system of patients presenting with chronic neck pain (including some with pain following whiplash injury) (Hagström and Carlsson, 1996; Sheather-Reid and Cohen, 1998; Koelbak Johansen et al., 1999).
The aim of this study was to determine whether such a condition is present in a larger group of patients fulfilling the criteria for ‘late whiplash syndrome’. As one of the authors had previously observed that sustained vibration at 100 Hz (delivered by a commercially available appliance) was perceived as a painful stimulus by some sufferers of chronic neck pain, it was decided to use this modality of sensory testing, in addition to standard tests of cutaneous sensory function.
Materials and Methods
Subjects
The inclusion criteria were based on the classification adopted by the Quebec Task Force (Spitzer et al 1995). Patients in Classes I and II with symptoms of more than six months duration subsequent to a motor-vehicle accident (MVA) were selected for the study. Patients with discernible musculoskeletal disorders, objective neurological deficits, or obvious psychopathology were not included in the study. The patient sample was not biased to either age or sex. A control group consisted of age and sex-matched healthy volunteers with no complaints of spinal, upper or lower limb pain or of injuries to the head, shoulder or knee regions necessitating medical treatment or time off work in the 12 months prior to testing.
All participants in the study were unpaid volunteers, living within the Perth metropolitan area and unconnected with this study. They had been referred from pain clinics and selected private practices in rheumatology and physiotherapy. All had given their informed written consent to a protocol approved by the Ethics Committee of Curtin University. Patients were allowed to continue taking their usual medication. It was not considered ethical or practicable to insist on a “wash-out” period of 24 hours before sensory testing, as many patients complained of pain that interfered with their ability to carry out normal daily activities.
Data Collection
Prior to psychophysical testing, demographic and health-related questionnaires were administered to all participants. In addition, specific accident-related information and symptomatology as recollected from acute phase were collected from the whiplash group. The Symptom Check List-90-R (SCL-90-R) (Derogatis, 1994) was used to assess psychological status of all participants. The Neck Disability Index (NDI) (Vernon, 1996) was used to provide an assessment of the extent of perceived functional disability in the whiplash patients.
A structured interview was held with each whiplash patient just prior to testing in order to document on a body chart the distribution of any current pain, and/or other symptoms. This information was compared with that obtained by the questionaires dealing with post-injury symptomatology. The Shortform McGill Pain Questionnaire (Melzack, 1987) was used to provide scores for sensory and affective pain descriptors, as well as intensity ratings of the overall pain experience, both currently and that recalled immediately after the accident. During the initial interview, great care was taken to ensure the comfort of patients and to allay any anxiety they may feel as a result of taking part in the study. Psychophysical testing was performed in an air-conditioned environment.
All sensory tests were performed by a single individual. Blinding of the investigator had been considered during the design of the study. However, it was decided to simulate, as far as possible, the normal clinical situation. Instructions given to each participant were standardized and care was taken not to provide information about expected test outcomes or about the research hypothesis.
Experiment 1: Qualitative psychophysical testing
In each participant, four standard cutaneous stimuli were used (Asbury, 1998). The aim was to stimulate different types of peripheral receptors using light-touch, punctate pressure, non-noxious cold and heat and to obtain a descriptive interpretation of stimulus quality by the participants. The stimuli were applied in a random order to the skin over the belly of the upper trapezius muscle on either side and over the central sternum. Each participant was asked to identify the presence and the quality of the stimulus for each area. Any differences in reported sensation between the areas were noted. For the thermal stimuli the participants were also instructed to rate the intensity of any painful sensation on a numerical rating scale from 0 to 100.
To elicit a sensation of light-touch, a cotton ball was fixed to a clamp and gently stroked four times across each test site. Touch pressure was applied over each site through a hand held Semmes-Weinstein monofilament (size 6.45). Two metal probes were used to assess thermal sensibility (Hot/Cold Discrimination kit with calibrated thermometers, Smith & Nephew Roylan Inc.). The cold probe was placed in ice and water (0°C) and the heat probe in warm water (40-41°C). The respective probes were maintained at a constant temperature. During application of heat and cold stimuli, the skin was touched obliquely with the rounded end of the metal probe for five seconds. The pressure applied was insufficient to indent the underlying skin.
Experiment 2: Quantitative psychophysical testing
Measurements were performed using a purpose built vibrameter with adjustable frequency (0-100Hz) and amplitude (0-2.5mm - peak to peak). The vibrating element produces pure sine waves that are transmitted by a small element, emerging from a supporting base. The possible influence of different application pressures on the accuracy of the readings is thereby minimised when the instrument is gently placed over soft tissues. A LED display indicates the frequency and amplitude of the delivered vibratory stimuli. The device was calibrated prior to testing and it was found to be accurate between 0.02-0.04mm for amplitude and between 1-2Hz for frequency. The resolution of the instrument was specified as being below 0.04mm for amplitude and ± 1Hz for frequency.
The vibration device was clamped to the end of an articulated arm, which was fixed to a portable trolley, thus ensuring stable application of the stimulus. Vibration was applied to skin over three test sites on each side of the body: the belly of the upper trapezius, the belly of the deltoid muscle, and the belly of the quadriceps muscle.
Different pre-determined test patterns were used to ensure that the stimulation sequence over the six test sites was conducted in a random manner. Randomization was necessary to exclude any potential reporting bias due to testing within or outside the area of described pain. From the available literature, it was unclear whether greater mechanical stimulation would result from increases in vibration frequency or in amplitude. Therefore, stimulation at each test site commenced alternately either with increasing frequency or amplitude in an ascending manner.
Vibration perception threshold (VPT) was determined by the subject operating a buzzer when first becoming aware of the stimulus. Amplitude or frequency readings were then recorded. The stimulus was further increased, either to a frequency of 100Hz, or to an amplitude of 1.0mm, whilst the other modality was kept constant at 0.2mm or 20Hz respectively. The participants were instructed to again sound the buzzer at the onset of any vibration-induced pain (VIP), thus providing an assessment of pain threshold. The location and the character of this pain were recorded on a body chart. If the participant was in pain prior to commencement of vibration testing, the buzzer was to be sounded if the intensity of pain increased or if pain spread into a ‘new’ body area. Unless there had been intolerance to submaximal stimulation, maximum vibration frequency or amplitude was maintained for 30 seconds at each test site.
Statistical methods
Descriptive statistics, normality testing (Lilliefors statistic) and Mann-Whitney-U test or 2-tail unpaired t-test were applied to cutaneous and vibration test results, and to the questionnaire. Pearson’s correlation coefficient or Spearman’s rank order correlation (rho) were used to assess the relationship between functional, psychological and pain scores. The significance level for all parametric/non-parametric tests was set at p<0.05. A logistic regression model evaluated the prognostic value of post-injury symptomatology, self-reported pain intensity with thermal stimuli and questionnaire scores in order to predict the presence of VIP in the patient group.
Reliability testing (Wilcoxon Signed Rank test and Spearman Rank Correlation) was conducted for the VPT/VIP in a subset of 16 healthy control participants and in the first 11 whiplash patients who had responded with VIP and agreed to undergo repeat testing.
Results
Demographic details
Forty-three patients, comprising 15 males and 28 females, of mean age 37 years (range 12-66 years), complained of neck and upper back pain of more than 6 months duration (median 18 months) following their involvement in a MVA. The commonest type of MVA were rear-end collisions (56%), followed in frequency by frontal (16%) and side-on (16%) collisions. Forty-three control subjects who were age and gender matched with the whiplash sample participated in the study.
At the time of their accident, 34 patients had been employed. Of these, 25 had taken on average 12-13 days off work, 6 had changed their occupation as a result of the accident and 8 had not returned to work. All had received ongoing medical treatment, which consisted mainly of analgesics, non-steroidal anti-inflammatory drugs and various modalities of physical treatment. Twenty-eight patients were involved in ongoing litigation, ten had settled their claims and five had never sought compensation for personal injury.
Symptomatology
Comparing the subjective reports, the intensity of neck pain and the presence of concentration problems appear to have remained constant since the accident. The incidence of headache tended to have declined, whereas that of referred pain (into shoulders and arms) to increase slightly with the passage of time. Other symptoms were rated as being less frequent with the passage of time (Table 1).
Table 1. – Self-reported symptoms acute (<2 weeks) and chronic (>6 months)
| Symptom | Acute | VAS Scores | Chronic | VAS Scores | ||||
| n | % | Mean | SD | n | % | Mean | SD | |
| Neck Pain | 43 | 100% | 82 | 15 | 43 | 100% | 37 | 15 |
| Headache | 43 | 100% | 77 | 21 | 38 | 88.4% | 74 | 19 |
| Shoulder Pain | 38 | 88.4% | 64 | 32 | 41 | 95.3% | 61 | 18 |
| Arm Pain | 26 | 60.5% | 39 | 37 | 25 | 58.1% | 59 | 22 |
| Pins and Needles | 22 | 51.2% | - | - | 19 | 44.2% | - | - |
| Numbness | 17 | 39.5% | - | - | 10 | 23.3% | - | - |
| Dizziness | 23 | 53.5% | - | - | 4 | 9.5% | - | - |
| Blurred Vision | 15 | 34.8% | - | - | 6 | 14.3% | - | - |
| Tinnitus | 13 | 30.9% | - | - | 1 | 2.4% | - | - |
| Concentration | 24 | 55.8% | - | - | 24 | 55.8% | - | - |
Pain distribution
At the time of testing, patients most commonly reported pain in the neck, shoulders, scapulae, and head. Next, in descending order of frequency, were the dorsal upper arms, lumbar region, interscapular region, thoraco-lumbar junction, and posterior thigh. Leg pain only occurred in those complaining of widespread spinal pain. Twenty-three patients reported either continuous or intermittent paraesthesiae, described as numbness and/or ‘pins and needles’, most frequently felt in their hands (dermatomes C6/7/8).
Qualitative psychophysical testing
All participants were able to perceive and correctly identify each of the four cutaneous stimuli. One patient reported that light touch over the pain-dominant shoulder was painful. Four patients rated punctate pressure applied in the region of their shoulder pain as painful. Eleven patients described a painful response to heat and twenty-three to cold stimulation. In the control group no participant reported pain in response to light-touch or punctate pressure, but ten experienced cold stimulation as painful and nine reported heat as painful. However pain felt on thermal stimulation was self-rated on a numerical scale as being 2-3 times higher in the whiplash patients (34/100 for heat, 29/100 for cold) than in the controls (14/100 for heat, 10/100 for cold).
Quantitative psychophysical testing
In all participants VPT was reported in the first 10-15% of available frequency and in the first 9-14% of available amplitude range. Normality testing was conducted on all perception threshold data collected from each individual test site. The results were positively skewed. Mann-Whitney U-tests showed no significant difference in vibration perception threshold between patients and control participants at the p<0.05 level. No significant differences in VPT were found between pre-test painful and non-painful test sites (Table 2).
Table 2. - Comparisons for VPT between whiplash and control group
| Groups° | n 1 W |
n2 C |
Test Point | Mann-Whitney U |
Significance 2-tailed p |
| Frequency Measurements | |||||
| W vs C | 43 | 43 | Knee | 886 | 0.73 |
| 43 | 43 | Trapezius | 817.5 | 0.35 | |
| 43 | 43 | Deltoid | 870 | 0.63 | |
| Amplitude Measurements | |||||
| W vs C | 43 | 43 | Knee | 762.5 | 0.16 |
| 43 | 43 | Trapezius | 764 | 0.16 | |
| 43 | 43 | Deltoid | 729.5 | 0.09 | |
°Groups: W=Whiplash patients, C= Control participants,
Twenty-eight whiplash patients (19 females and 9 males) reported VIP. In 13 patients the stimulus had to be removed before the predetermined maximal intensity and time of application were reached. Although the onset of both VIP and pain intolerance was reported through a wide range, the median of responses occurred in the last quarter of available amplitude and frequency stimulus intensity at all three test sites (Table 3). Most patients reported VIP with an increase in both modalities at the same test site. Two patients reported pain only with an increase in frequency, whilst one patient only to an increase in amplitude. No control subject reported VIP or any discomfort due to vibration.
The majority of pain responses in the patients occurred at the trapezius test site, when frequency (n=25) or amplitude (n=26) was increased. Stimulation over the deltoid muscle belly produced painful responses, both with increases in amplitude (n=23) and in frequency (n=18). In these patients, vibration over both regions reproduced their pre-existing pain. Fourteen patients reported pain spreading into ‘new’ areas. Some patients reported VIP at the knee test site (10 with increasing amplitude and 8 with increasing frequency). In these patients, vibration evoked pre-existing pain, presumably referred from the low back into the lower limb. In eight patients VIP was recorded at all three test sites. Onset of vibration induced pain at any one site was considered a positive response.
The non-painful responses to vibration elicited from 16 healthy controls and the VIP in 11 patients were reproducible in a test/retest context, with at least two weeks separating the two tests.
Table 3. Vibration induced pain threshold and tolerance
| n | Knee median (range) |
Trapezius median (range) |
Deltoid median (range) |
|
| Amplitude | ||||
| Pain Threshold | 28 | 1.0 (0.42-1.0) µm | 0.72 (0.32-1.0) µm | 0.96 (0.36-1.0) µm |
| Pain Tolerance | 11 | 1.0 (0.78-1.0) µm | 0.90 (0.68-1.0) µm | 0.91 (0.68-1.0) µm |
| Frequency | ||||
| Pain Threshold | 26 | 100 (27-100) Hz | 84 (22-100) Hz | 82 (28-100) Hz |
| Pain Tolerance | 12 | 72 (42-100) Hz | 87 (30-100) Hz | 83 (56-100) Hz |
Scores on self-reported questionnaires
The patients presented with high scores in all five components of the Shortform McGill pain questionnaire. Mann-Whitney U-test results showed that those who reported VIP selected significantly more affective pain descriptors than those without VIP.
As a group, the patients scored a mean of 44.8% (SD 14.9%) of functional impairment on the NDI, which is rated as a ‘severe’ level of disability (40-60%) (Vernon 1996). Pain was the dominant problem, but travelling, activities of social life, personal care, sexual activity and sleep were also disrupted. No significant difference in the NDI was found between patients who were involved in litigation and those who had settled their claims or had never claimed compensation for personal injury. No significant difference in NDI was found between patients with and without VIP.
Forty-one patients and 42 control subjects completed the psychological questionnaire. As two of the participants were under 14 years of age, the adult questionnaire could not be used. One whiplash patient declined to complete the questionnaire. The SCL-90-R results revealed an overall elevated level of distress in the patients compared to the controls. Significant differences between patients and controls were found in the dimensions of somatisation, obsessive-compulsive, depression, anxiety, hostility and the three global indexes (GSI, PSDI, PSI) (Figure 1). The presence of VIP was not predictive of higher levels of psychological distress.
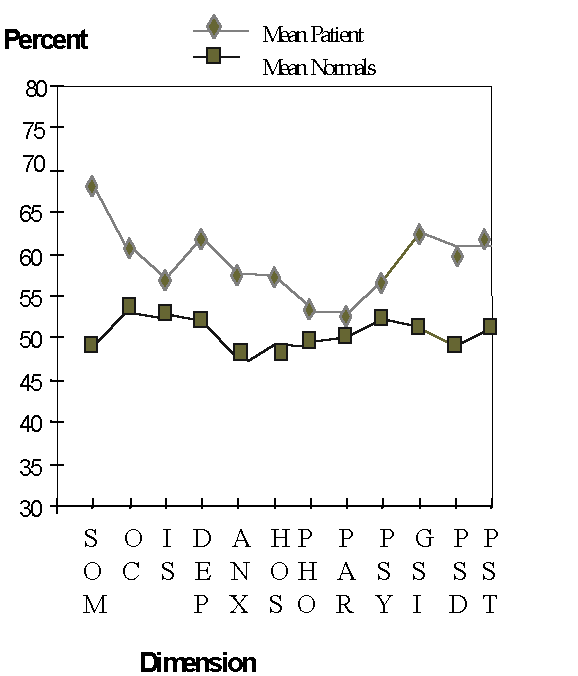
Fig.1 – SCL-90-R Clinical profile for study groups
SOM= somatisation, 0-C= obsessive-compulsory, I-S= interpersonal sensitivity, DEP= depression, ANX= anxiety, HOS= hostility, PHO= phobic-anxiety, PAR= paranoid ideation, PSY= psychoticism, Global Indexes: GSI= Global Severity Index, PST= Positive Symptom Total index, PSD= Positive Symptom Distress index
Correlations
All nine primary symptom dimensions and three global indexes on the SCL-90-R were analysed for correlation with the NDI and the Shortform McGill pain questionnaire in the whiplash group (n=41), using Pearson’s and Spearman’s Rank Order Correlation. In the whiplash group, there was high positive correlation for the NDI with most variables of the Shortform McGill pain questionnaire (Table 4). There was no significant correlation between any of the psychological dimensions of the SCL-90-R and the NDI. Spearman’s Rank Order Correlation showed significant correlation between the VAS of the McGill pain questionnaire with the scores in the psychological dimension ‘somatisation’ (r=0.32, p=0.02) and the ‘PSDI-global index’ (Positive Symptom Distress Index) (r=0.35, p=0.02) of the SCL-90-R.
Regressions
A logistic regression analysis was conducted to identify the variables that might predict either the presence or absence of VIP in a patient with chronic pain following a whiplash injury. Due to the small sample size, different combinations of variables were tested with regression models. Only the intensity of arm pain post-injury appeared to be significant (p<0.05). Factors which tended to correctly predict group membership were the presence of paraesthesiae post-injury, high affective pain descriptor scores (Shortform McGill pain questionnaire) and self-reported pain intensity with heat stimuli (Table 5).
Table 4. Nonparametric Correlations - Spearman’s rho (n=43)
| MG-S | McG-A | McG-T | VAS | ||
|---|---|---|---|---|---|
| NDI | CC | .27 | 0.36b | 0.34b | 0.61a |
| Sig. | .08 | 0.02 | 0.03 | 0.00 |
CC= Correlation Coefficient, Sig.= Significance (2-tailed) McG-S = sensory dimension; McG-A = affective dimension; McG-T = total pain descriptors; VAS = visual analogue scale a significant (p<0.01) b significant (p<0.05)
Table 5. Backward Stepwise (LR) regression analysis
| Variable | B | SE | Sig | Exp (B) | 95% CI | Predictive Power % |
|---|---|---|---|---|---|---|
| Arm pain | 0.03 | 0.01 | 0.03* | 1.03 | 1.00-1.05 | 70.3 |
| McGill affective | 0.25 | 0.14 | 0.06 | 1.29 | 0.99-1.68 | 72.1 |
| Heat mean | 1.51 | 0.89 | 0.09 | 4.51 | 0.78-25.96 | 72.1 |
| Paraesthesiae | 0.96 | 0.71 | 0.18 | 2.61 | 0.65-10.56 | 70.0 |
| Depression | 0.25 | 0.13 | 0.05 | 1.28 | 0.99 - 1.65 | 72.9 |
The principal findings
The principal finding was that 65% of patients in the sample reported VIP. Those with VIP were more likely to report referred pain and paraesthesiae into the upper limb, both soon after the MVA, as well as at time of testing. Furthermore, stimulation with cold and heat elicited a proportionately higher number of pain responses in these patients. Although this group used affective pain descriptors more frequently, neither a significantly higher NDI nor higher psychological scores were found compared to the other patients.
Discussion
Although the information collected regarding accidents was retrospective, the demographic and clinical characteristics of the patient sample were similar to those reported by others (Barnsley et al., 1994; Radanov et al., 1995; Panjabi et al., 1998). The relatively high proportion of patients with upper limb pain (58%) and paraesthesiae (44%) was unexpected. There is clinical evidence suggesting that these are referred symptoms of cervical neural origin (Quintner, 1989).
Experimental findings
Cutaneous vibration sense is mediated through specialised mechanoreceptors which respond to different frequencies - Merkel’s discs respond to frequencies between 5-15 Hz, Meissner’s corpuscles to frequencies between 20-50 Hz and Pacinian corpuscles to frequencies between 60-400 Hz (Kandel et al., 2000). The vibration parameters used in this study were sufficient to elicit responses from each type of receptor. There were no significant differences in VPT between the patients and the control subjects. Furthermore, testing conducted within an area of pain did not appear to have any significant influence on VPT. The unimpaired VPT in the patients is indicative of normal mechanoreceptor and large sensory fibre function. Our findings confirm that VPT tends to occur early in the available stimulus range (first 20-25%) and that skewed results are to be expected (Bloom et al., 1984; Yarnitsky, 1996). The applied ‘method of limits’ for threshold detection does show large inter-sessional bias as discussed by Yarnitsky (1996) and may to some extent be dependent on subject’s attentional and cognitive factors (Snodgrass et al., 1985; Schwartz and Klima 1995).
A region of secondary hyperalgesia is characterised by a change in the modality of the sensation evoked by low threshold mechanoreceptors, from touch to pain, and an increase in the magnitude of pain sensations evoked by mechanically sensitive nociceptors (Lamotte et al., 1991; Cervero et al., 1994). In the majority of patients, VIP occurred in the last quarter of available stimulus range. Increasing the amplitude of the vibration stimulus, is known to increase the total number of active sensory neurons in a linear fashion (Kandel et al., 2000). Presumably, the resulting spatial summation of spike trains was sufficient to discharge sensitized second order sensory neurones. The similar response seen when the frequency of vibration was increased can be explained by the phenomenon of temporal summation of afferent impulses. These findings imply that VIP following whiplash injury is a clinical correlate of central sensitization. Furthermore, the spread of VIP into ‘new’ areas, as observed in 14 patients, is consistent with such a state of heightened sensibility of the central nervous system (Coderre et al., 1993; Woolf and Mannion, 1999).
The distribution of chronic pain was diverse in the patient population and does seem to have influenced the responses to vibration at the different test sites. Increased stimulus intensity also means an increase in the length of application time. Further research will be necessary to determine whether the test body site, the duration of application or its intensity that are critical factors determining response to vibration.
Because of the proximity of the trapezius test site to the cervical spine, cutaneous vibration over this region might have been conducted to receptors located in putative occult injuries of deep cervical spinal structures, one candidate being zygapophyseal joints (Wallis et al., 1997). In this scenario, VIP could reflect peripheral sensitization of nociceptors related to these joints.
VIP occurring in eight patients at all three test sites raises the possibility of hypervigilance, manifested by a generalised increase in sensitivity to experimentally induced stimulation (McDermid et al., 1996). The negative cognitive and emotional experiences of individual patients might have amplified vibration at the supraspinal level to the extent that it became an aversive stimulus. However, as the cognitions and beliefs of individual patients were not explored in this study, this possibility cannot be refuted.
The number of patients reporting pain to mechanical cutaneous stimuli was low and no conclusion can be drawn from it. However, it was of interest that in the same patients, vibration was also reported as painful. While both findings demonstrate mechanically induced pain (allodynia or hyperalgesia), it is possible that different underlying pain mechanisms may exist amongst the group of positive responders to vibration. As such, four patients rated punctate pressure exerted by the Semmes-Weinstein monofilament in the area of their shoulder pain as painful. Three of them also reported pain in response to heat and cold over the same site. In these patients, all stimuli appear to have been of sufficient intensity to activate polymodal small diameter fibres (Ochoa and Yarnitsky, 1993; Magerl et al., 1998, Pederson and Kehlet, 1998).
Mechanical stimulation caused by the application pressure of the thermal probes was considered to have been minimal. Moderate heat, expected to elicit a sensation of ‘warmth’ rather than pain, was rated as being painful by 9 of 43 control participants. Such responses were difficult to explain. However, the evoked pain intensity ratings were significantly lower than those of 11 patients, whose reported ratings could therefore be classed as hyperalgesic and mediated by a small group of polymodal high-threshold afferents (Cesare and McNaughton, 1997; Morin and Bushnell, 1998). The blinding to the heat stimulus might have resulted in heightened awareness in all participants???, causing them to be unduly hypervigilant in this situation.
The short duration of application of the cold stimulus appears to have activated both innocuous cold receptors (Ad-fibres) and polymodal nociceptors (Davis, 1998). Ten control participants reported pain in response to cold, implying a normal discharge response of some polymodal units (Davis, 1998). Five out of those 10 responses were recorded at the sternal test site where lack of subcutaneous fat the site might have contributed to the responses. The painful responses to cold in 23 patients can be attributed to responses from the same receptor types. Cold hyperalgesia is prevalent in patients with neuropathic pain (Ochoa, 1996; Davis, 1998). The higher proportion of painful responses seen in the whiplash group is consistent with this phenomenon.
Psychological profiles and disability
The patients exhibited an enhanced level of psychological distress across all dimensions of the SCL-90-R, when compared to the controls. The finding of significantly elevated scores for the dimensions of somatisation, obsessive-compulsive behaviour, depression, anxiety and hostility is consistent with the findings of others (Wallis et al., 1996; Wallis et al., 1997; Wallis et al., 1998; Kessels et al., 1998). This relatively homogeneous psychological profile is thought to be the result of the chronic pain state and not a reflection of primary psychopathology (Wallis et al., 1996). The significant positive correlation found in this study between the VAS pain intensity scale (Shortform McGill pain questionnaire) and both the somatisation dimension and ‘positive symptom distress index’ of the SCL-90-R, supports there being a relationship between ongoing pain and distress arising from bodily dysfunction.
Patients who reported VIP had significantly higher affective but not sensory pain descriptor scores on the Shortform McGill pain questionnaire. Affective pain descriptor scores were also significantly correlated with scores on the NDI. Other studies have shown that high affective scores tend to correlate with higher ratings of distress, and that there is a positive association between distress and self-reported functional disability (REF!!)
It has been suggested that psychosocial factors may be major risk factors for development of the late whiplash syndrome (Smed, 1997). The medico-legal process has been identified as one such factor that can reinforce and perpetuate maladaptive pain behaviour (Shapiro and Roth, 1993; Swartzman et al., 1996; Schmand et al., 1998; Cassidy et al., 2000). Therefore, as mentioned above, some patients with outstanding medico-legal claims and/or with high levels of psychological distress may be biased in favour of reporting painful responses during quantitative sensory testing (McDermid et al., 1996). In this study, no significant difference was found between patients with and without VIP for any dimension of the SCL-90-R. The 28 patients with VIP (13 being intolerant to the stimulus) did not differ from the 15 without VIP with respect to the status of their personal injury claims. Furthermore, patients pursuing personal injury claims did not rate themselves as being significantly more disabled (NDI) than those without ongoing litigation. Although the numbers are small, these findings suggest that the reported responses to vibratory stimuli were independent of the psychological and litigation status of the patients.
Conclusions
Patients presenting with the late whiplash syndrome appear to be a heterogeneous group in terms of underlying pain mechanisms. This cross-sectional sample contained those who processed vibratory stimuli in a manner consistent with the phenomenon of central sensitization. Areas of widespread pain reported by these patients are therefore likely to represent regions of secondary hyperalgesia, raising a number of important clinical implications.
Firstly, the clinical features can be rationally explained in a neurobiological framework. This has not proved possible when using the standard ‘injury’ model, nor with the model of primary psychogenesis. Secondly, it predicts that mechanical forms of passive therapeutic intervention will be ineffective, and possibly harmful. Thirdly, the increased level of psychological distress, a higher tendency to use affective pain descriptors and a high level of perceived disability in the whiplash group, predicates that the approach to pain management in such chronic pain patients should be multidimensional.
Acknowledgments
The authors wish to thank Mr. P. Kolb (Bioengineering Division, Royal Perth Hospital), who designed and constructed the vibration device for this study, Mr. K. Sussenbach for technical assistance, Ms L. Lester and Dr. J. Sommer for statistical advice, Dr. P. Siddall and Prof. M. Cousins for their helpful suggestions.
References
Asbury A. Numbness, tingling, and sensory loss. In: Fauci AS, Braunwald E, Isselbacher KJ, Wilson JD, Martin JB, Kasper DL, Hauser SL, Longo DL, editors. Harrison’s Principles and Practice of Medicine. 14th ed. New York: McGraw Hill, 1998: 122-125.
Barnsley L, Lord SM, Bogduk N. Whiplash injury. Pain 1994;58:283-307.
Bloom S, Till S, Soenksen P, Smith S. Use of biothesiometer to measure individual vibration thresholds and their variation in 519 non-diabetic subjects. Brit Med J 1984; 288:1793-1795.
Cassidy JD, Carroll LJ, Cote P, Lemstra M, Berglund A, Nygren A. Effect of eliminating compensation for pain and suffering on the outcome of insurance claims for whiplash injury. NEJM 2000; 342:1179-1186.
Cervero F, Meyer RA, Campbell JN. A psychophysical study of secondary hyperalgesia: evidence for increased pain to input from nociceptors. Pain 1994;58:21-28.
Cesare P, McNaughton P. Peripheral pain mechanisms. Curr Opin Neurobiol 1997;7:493-499.
Coderre TJ, Katz J, Vaccarino AL, Melzack R. Contribution of central neuroplasticity to pathological pain. Pain 1993;52:259-285.
Davis KD. Cold-induced pain and prickle in the glabrous and hairy skin. Pain 1998;75:47-57.
Derogatis LR. Symptom Checklist-90-R. Administration, scoring and procedures manual. Minneapolis, National Computer Systems Inc,1994.
Devor M. The pathophysiology of damaged peripheral nerves. In: Wall PD, Melzack R, editors. Textbook of Pain. Edinburgh: Churchill Livingstone, 1994. pp. 79-100.
Ferrari R, Russell AS. Epidemiology of whiplash: an international dilemma. Ann Rheum Dis 1999a;58:1-5.
Ferrari R, Russell AS. Development of persistent neurologic symptoms in patients with simple neck sprain. Arthritis Care Res 1999b;12:70-76.
Gargan M, Bannister G, Main C, Hollis S. The behavioural response to whiplash injuries. J Bone Joint Surg 1997;79B:523-526.
Hagström Y, Carlsson J. Prolonged functional impairments after whiplash injury. Scand J Rehab Med 1996;28:139-146.
Kandel ER, Schwartz JH, Jessell TM, editors. Principles of Neural Science. 4th ed. New York: McGraw Hill, 2000: 430-450.
Kessels RP, Keyser A, Verhagen WI and van Luijtelaar EL. The whiplash syndrome: a psychophysical and neuropsychological study towards attention. Acta Neurol Scand 1998;97:188-193.
Koelbaek Johansen M, Graven-Nielsen T, Schou Olesen A, Arendt-Nielsen L. Generalised muscular hyperalgesia in chronic whiplash syndrome. Pain 1999;83: 229-234.
Koltzenburg M, Lundberg LE, Torebjoerk HE. Dynamic and static components of mechanical hyperalgesia in human hairy skin. Pain 1992;51:207-219.
Koltzenburg M, Torebjoerk HE, Wahren LK. Nociceptor modulated central sensitization causes mechanical hyperalgesia in acute chemogenic and chronic neuropathic pain. Brain 1994;117:579-591.
Lamotte RH, Shain CN, Simone DA, Tsai E-FP. Neurogenic hyperalgesia: psychophysical studies of underlying mechanisms. J Neurophysiol 1991; 66: 190-211.
Magerl W, Wilk SH, Treede RD. Secondary hyperalgesia and perceptual wind-up following intradermal injection of capsaicin in humans. Pain 1998;74:257-268.
McDermid AJ, Rollman GB, McCain GA. Generalized hypervigilance in fibromyalgia: evidence of perceptual amplification. Pain 1996;66:133-144.
Melzack R. Neurophysiological foundations of pain. In: Sternbach RA, editor. The psychology of pain. 2nd ed. New York: Raven Press, 1986: 1-23.
Melzack R. The short-form McGill pain questionnaire. Pain 1987;30:191-197.
Merskey H, Bogduk N, editors. Classification of chronic pain. Description of chronic pain syndromes and definitions of pain terms. 2nd ed. Seattle: IASP Press, 1994: 209-213.
Morin C, Bushnell MC. Temporal and qualitative properties of cold pain and heat pain: a psychophysical study. Pain 1998;74:67-73.
Obelieniene D, Schrader H, Bovim G, Miseviciene I, Sand T. Pain after whiplash: a prospective controlled inception cohort study. J Neurol Neurosurg Psychiatry 1999;66:279-283.
Ochoa JL, Yarnitsky D. Mechanical hyperalgesia in neuropathic pain patients: dynamic and static subtypes. Ann Neurol 1993;33:465-472.
Ochoa JL. Human polymodal receptors in pathological conditions. In: Kumazawa T, Krueger L, Mizumura K, editors. Progress in Brain Research. Amsterdam: Elsevier Science BV, 1996.113:185-197.
Panjabi MM, Cholewicki J, Nibu K, Babat LB, Dvorak J. Simulation of whiplash trauma using whole cervical spine specimens. Spine 1998;23:17-24.
Pearce JM. Whiplash injury: a reappraisal. J Neurol Neurosurg Psychiatry 1989;52:1229-1231.
Pearce JM. A critical appraisal of the chronic whiplash syndrome. J Neurol Neurosurg Psychiatry 1999;66:273-276.
Pederson JL, Kehlet H. Secondary hyperalgesia to heat stimuli after burn injury in man. Pain 1998;76:377-84.
Quintner JL. A study of upper limb pain and paraesthesiae following neck injury in motor vehicle accidents: an evaluation of the brachial plexus tension test of Elvey. Brit J Rheumatol 1989;28:528-533.
Radanov BP, Sturzenegger M, Di Stefano G. Long-Term outcome after whiplash injury. A 2-year follow-up considering features of injury mechanism and somatic, radiologic, and psychological findings. Medicine (Baltimore) 1995;74:281-297.
Radanov BP, Bicik I, Dvorak J, Antinnes J, von Schulthess GK, Buck A. Relationship between neuropsychological and neuroimaging findings in patients with late whiplash syndrome. J Neurol Neurosurg Psychiatry 1999;66:485-89.
Ratcliff AH. Whiplash injuries. J Bone Joint Surg 1997;79:517-519.
Schmand B, Lindebloom J, Schagen S, Heijt R., Koene T, Hamburger HL. Cognitive complaints in patients after whiplash injury: the impact of malingering. J Neurol Neurosurg Psychiatry 1998;64:339-343.
Schwartz B, Klima RR. Vibration sensation: measurement techniques and applications. Critical Reviews in Physical and Rehabilitation Medicine 1995;7:113-130.
Shapiro AP, Roth R. The effect of litigation on recovery from whiplash injury. Spine: State of the Art Reviews. Philadelphia: Hanley & Belfus, Inc. 1993;7:531-556.
Sheather-Reid RB, Cohen ML. Psychophysical evidence for neuropathic components of chronic neck pain. Pain 1998;75:341-347.
Smed A. Cognitive function and distress after common whiplash injury. Acta Neurol Scand 1997; 95:73-80.
Snodgrass JG, Levy-Berger G, Haydon M. Human Experimental Psychology. New York: Oxford University Press, 1985. pp: 56-87.
Spitzer WO, Skovron ML, Salmi LR, Cassidy JD, Duranceau J, Suissa S, Zeiss E. Scientific monograph of the Quebec Task Force on whiplash associated disorders: redefining "whiplash" and its management. Spine 1995;20:8S:10S-20S.
Swartzman LC, Teasell RW, Shapiro AP, McDermid AJ. The effect of litigation status on adjustment to whiplash injury. Spine 1996;21:53-58.
Vernon H. The neck disability index: patient assessment and outcome monitoring in whiplash. J Musculoskel Pain 1996;4:95-104.
Wallis BJ, Lord SM, Barnsley L, Bogduk N. Pain and psychologic symptoms of Australian patients with whiplash. Spine 1996;21:804-810.
Wallis BJ, Lord SM, Bogduk N. Resolution of psychological distress of whiplash patients following treatment by radiofrequency neurotomy: a randomised, double-blind, placebo-controlled trial. Pain 1997;73:15-22.
Wallis BJ, Lord SM, Barnsley L, Bogduk N. The psychological profiles of patients with whiplash-associated headache. Cephalalgia 1998;18:101-105.
Woolf CJ, Doubell TP. The patho-physiology of chronic pain - increased sensitivity to low threshold Aß-fibre input. Curr Opin Neurobiol 1994;4:525-534.
Woolf CJ, Mannion RJ. Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet 1999;353:1959-1964.
Yarnitsky D. Quantitative sensory testing. Muscle Nerve 1996;20:198-204.


